Vibrational and electrochemical studies of pectin - a candidate towards environmental friendly lithium-ion battery development
Dr. Phillip Wu, a visiting scholar at the Institute, together with his co-workers at the National Taipei University of Science and Technology, initiated and coordinated Dr. Yeng-Long Chen and Dr. Maw-Kuen Wu’s groups, and collaborated with Dr. Chia-Liang Cheng of National Dong-Hwa University, to complete a paper entitled “Vibrational and Electrochemical Studies of Pectin – A Candidate Towards Environmental Friendly Li-ion Battery Development”, which was recently accepted for publication on PNAS-Nexus. We believe this is the first paper by scientists working in Taiwan to appear in the PNAS-Nexus online, which was launched in March this year.
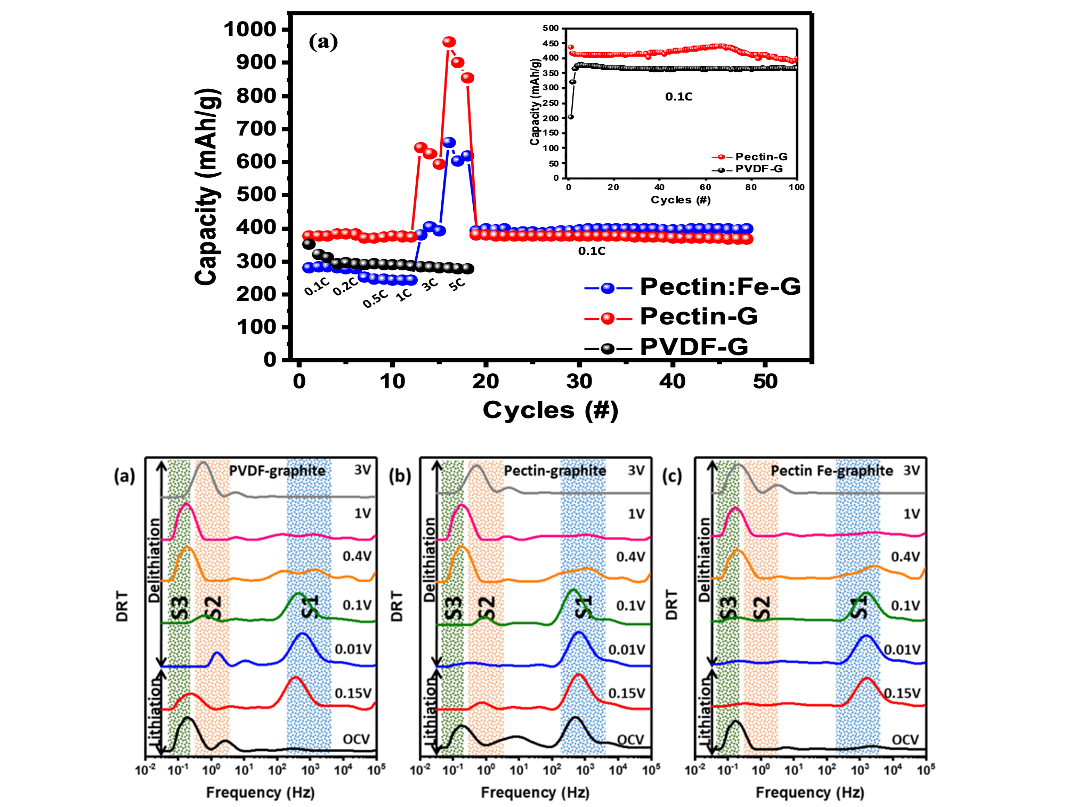
This work describes a methodology for creating pectin films, including both pristine pectin and Fe-doped pectin, and explores their potential for lithium-ion battery application. Fe doping enhances the conductivity observed in electrochemical impedance spectroscopy (EIS), and from the magnetic response of pectin evidence for Fe3+ is identified. The Li-ion half-cell prepared with pectin as binder for anode materials such as graphite shows stable charge capacity over long cycle life, and with slightly higher specific capacity compare with the cell prepared using PVDF as binder. A novel enhanced charging specific capacity at high C-rate is observed in cells with pectin binder suggesting within a certain rate (~5C) pectin-graphite anode exhibits higher capacity at faster charge rates, as shown in figure A. When placed in electrode it improves the material elastic properties, material inclusion and fundamentally affects the transport mechanisms. Combining Fe in pectin improves the capacity and maintains fast charging performance. This work demonstrates that pectin, due to its biodegradable and non-toxic nature, has emerged as an attractive material for Li-ion battery application for environmental friendly recycle processes.
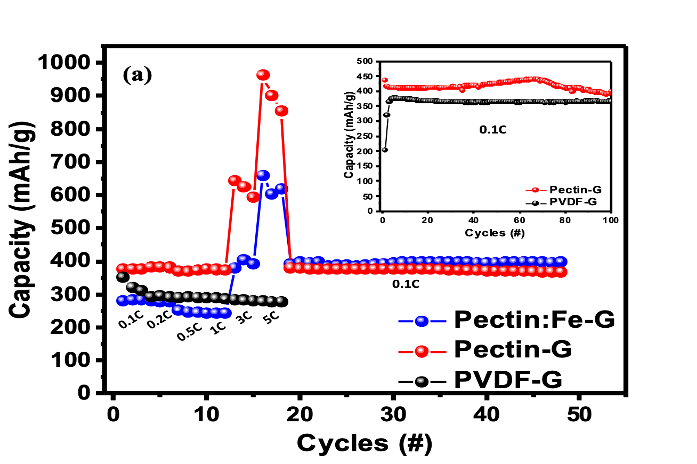
Figure 1. Specific capacities at different C-rate for graphite with PVDF, pectin and pectin:Fe binder. The inset is the long cycle test for PVDF and pectin binders. The results show that with pectin or pectin-Fe binders the specific capacities are enhanced at C-rate higher than 1C.
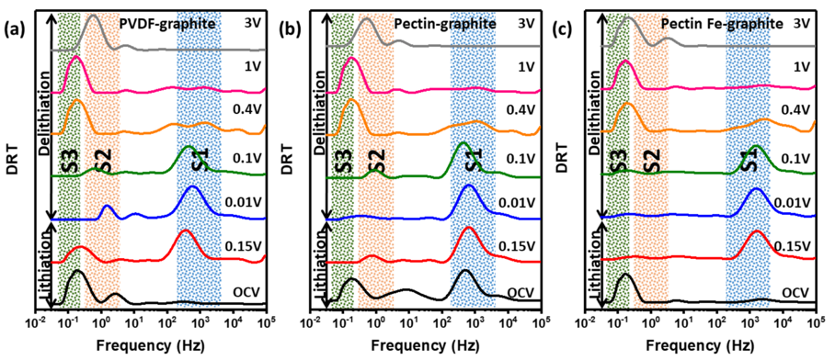
Figure 2. Electrochemical impedance spectroscopy (EIS) coupled with the distribution of relaxation times is employed to study the ion transport for all the samples at both lithiation and delithiation conditions. The positions of peaks S1, S2 and S3 correspond to surface film resistance, and electrode/electrolyte interfacial charge transfer resistances, respectively. The analysis confirms the lithiation process is binder induced. The total impedance is much lower for graphite-Fe-doped pectin sample indicating better conductivity and charge transfer mechanism. Overall, we can confirm that the presence of Fe facilitates the charge transfer process and the electronic conductivity.
Journal Links: https://doi.org/10.1093/pnasnexus/pgac127